|
The carbon-oxygen double bonds in #"CO"_2# molecules are polar, with carbon bonded to the second-most electronegative element on the periodic table. Carbon dioxide molecules contain no hydrogen atoms, so it should be safe to rule out the presence of hydrogen bonds. Hydrogen bonds are the interaction between hydrogen atoms bonded to Oxygen/Nitrogen/Fluorine atoms and lone pairs of electrons. There are three types of intermolecular interactions, as seen in the order of (typically) strongest to weakest: 
One last thing, hydrogen bonds are the strongest force out of the intermolecular forces but it is not the strongest when compared with other types of bonding such as covalent bond as covalent bonds are way stronger than hydrogen bonds! An example of hydrogen bond is water molecules. Hydrogen bonds are the strongest and occur when there is either a fluoride, oxygen or nitrogen molecule which is bonded with a hydrogen which is then joined with either a fluoride, oxygen or nitrogen molecule. Dipole-dipole forces are the attraction between the positive end of one polar molecule with the negative end of another polar molecule. Since #CO_2# is made of one carbon and 2 oxygen and both carbon and oxygen are non-metals, it also have covalent bonds.įor extra information, there are 3 types of intermolecular forces.ĭispersion forces are weaker than dipole-dipole and dipole-dipole are weaker than hydrogen bonds.ĭispersion forces are usually present in all molecules and are temporary. In this case, dipole-dipole intraction occur between this molecules because hydrogen have partial positive charge and Cl have partial negative charge, because Cl have more electronegative charge compare with hydrogen.ġ.#CO_2# has dispersion forces or van der waals forces as its only intermolecular force. Mainly, after intraction it is make N-H bonds, N-O bonds and N-F bonds. Mainly, Hydrogen bonding occur when hydrogen are attached with Nitrogen, oxygen and F. HCl is the best example of hydrogen bonding intermolecular forces. Mainly, hydrogen bonding occur on polar molecules. hydrogen bonding:- hydrogen bonding is a special type of intermolecular forces. this type of intraction is possible only on polar molecules. intraction between two saparate molecules, this is called dipole-dipole forces. So, the intraction between one molecules to another molecules is called dipole-dipole intraction. 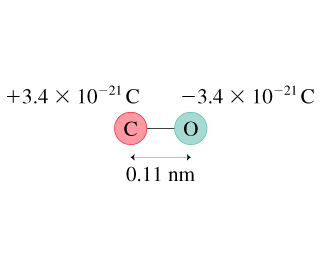
Intraction of one molecules to another molecules each molecules possess dipole moment. due to this, carbon have a partial positive charge and oxygen have negative charge. How you can say that CO is polar molecules?ĬO is a polar molecules because oxygen is highly electronegativity atoms compare with carbon. 
the dipole-dipole intraction between CO molecules are strongest then london dispersion forces. You know that, dipole-dipole intraction occur in polar molecules. but it all depends upon molecules and atom. so, we can say that, different molecules created different types of intermolecular forces such as, dipole dipole interaction, hydrogen bonding, ion dipole interaction, and London dispersion forces. CO is best example for force of attraction and repulsion between molecules. 
I think, You can understand with above defination, the force of attraction and repulsion between molecules or particle. Advertisements Intermolecular forces in CO?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
